EMA report on geographic distribution of clinical trials supports need for revision of European clinical trial legislation

What you Need and When – The Key Documents in the Drug Lifecycle - Trilogy Writing & Consulting GmbH

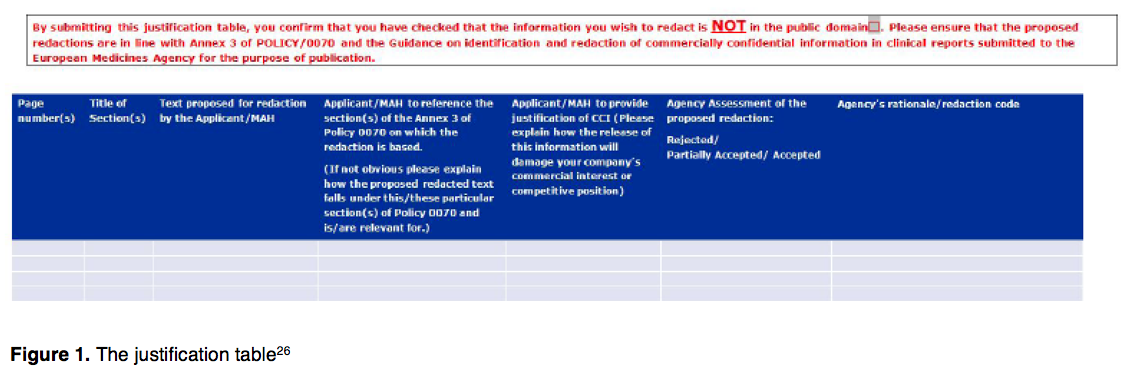
Positive outcome for ema's policy 0070, one year after the publication of the clinical trial results - Portolano Cavallo
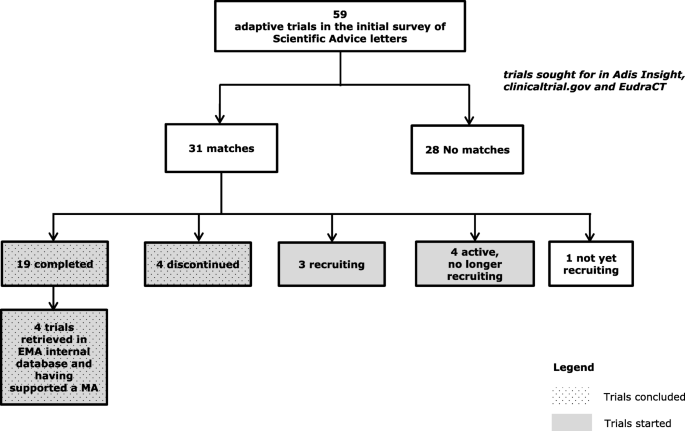
Adaptive designs in clinical trials: from scientific advice to marketing authorisation to the European Medicine Agency | Trials | Full Text
The European Medicines Agency Clinical Data Website Enables Insights Into Clinical Development Timelines And Strategy. - Document - Gale Academic OneFile
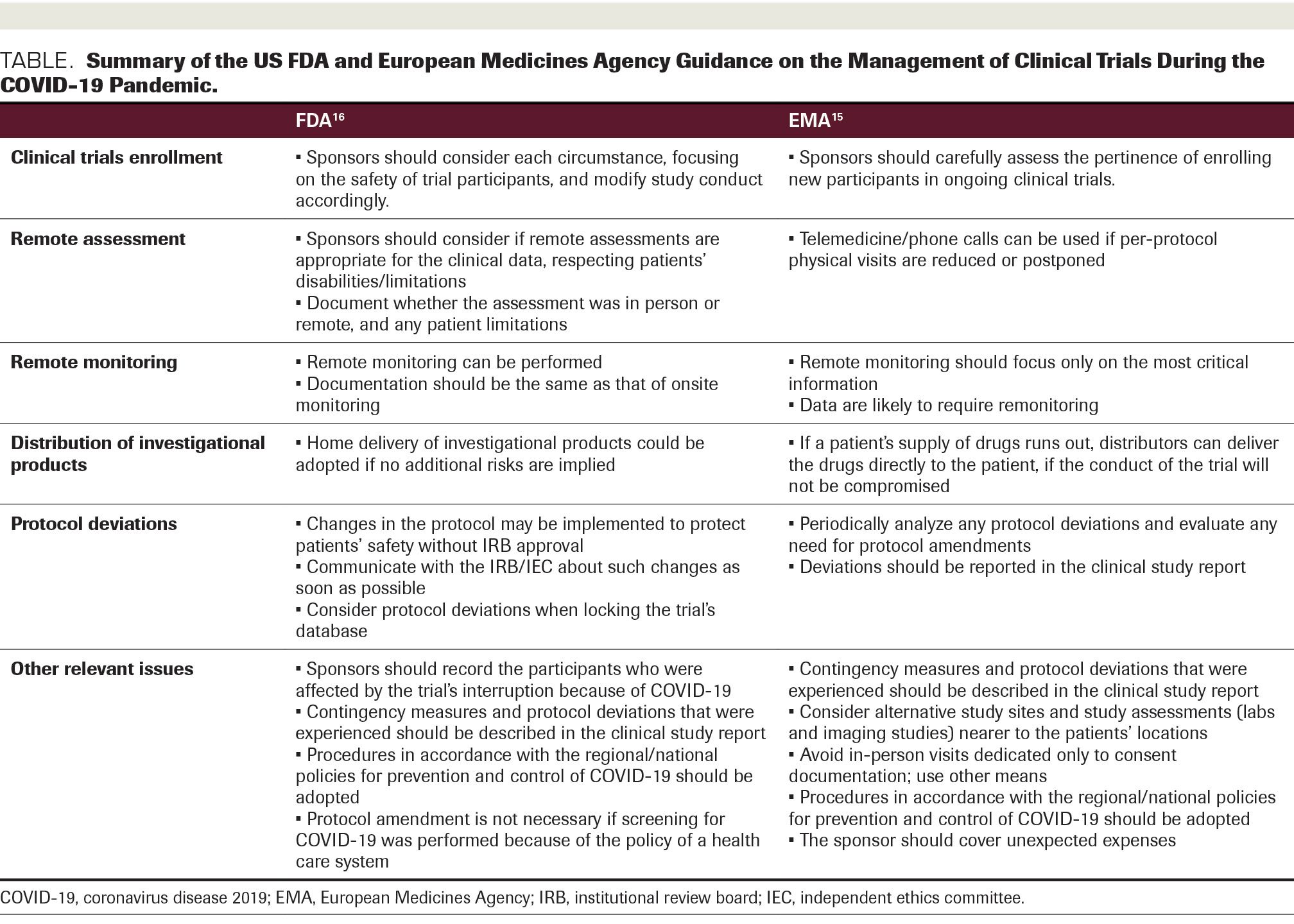
Transparency of Regulatory Data across the European Medicines Agency, Health Canada, and US Food and Drug Administration | Journal of Law, Medicine & Ethics | Cambridge Core

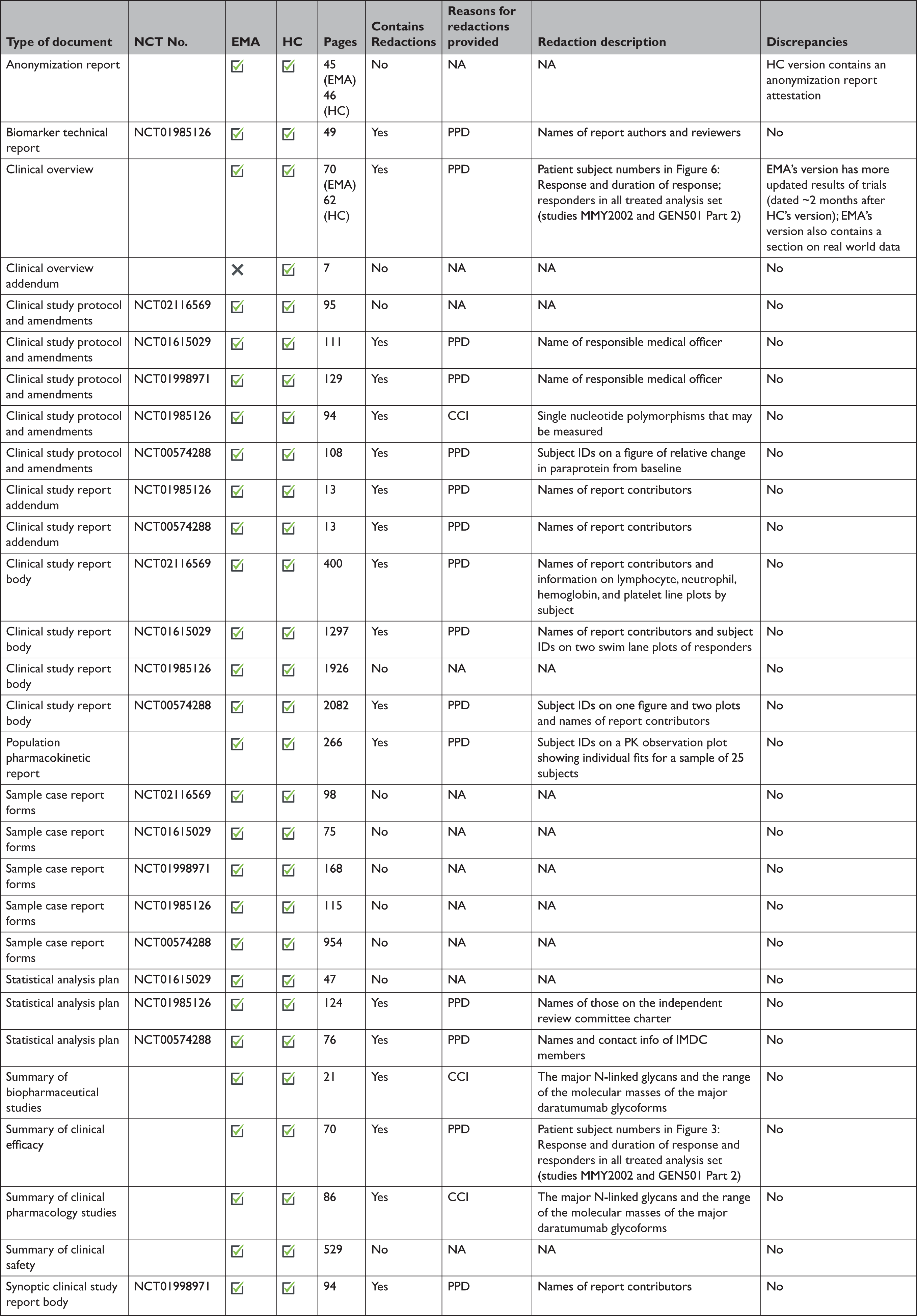
PDF) Evaluating the re-identification risk of a clinical study report anonymized under EMA Policy 0070 and Health Canada Regulations






![PDF] Effective authoring of clinical study reports: A companion guide | Semantic Scholar PDF] Effective authoring of clinical study reports: A companion guide | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2f8d38c9afbd7fce7a7635cf459a7989c82a753a/5-Table1-1.png)